
You cannot use the original one, because that would go against the flow of the lattice enthalpy arrow. 
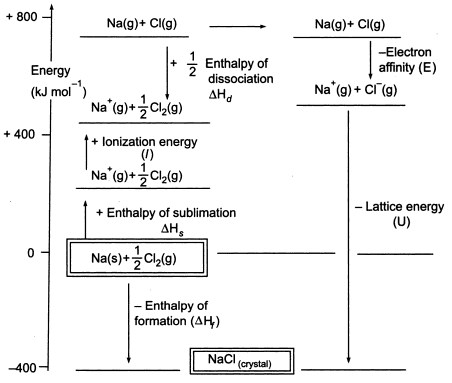
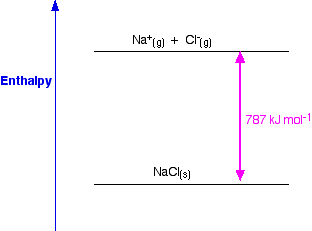
It does, of course, mean that you have to find two new routes. The only difference in the diagram is the direction the lattice enthalpy arrow is pointing. Let us begin with calcium chloride: as a crystal of CaCl 2 dissolves in water, interactions between ions are broken and new interactions between and water molecules and ions and. How would this be different if you had drawn a lattice dissociation enthalpy in your diagram? Your diagram would now look like this: Since both processes occur (all these compounds are highly soluble in water), the G’s for the formation of all three solutions must be negative. So, from the cycle we get the calculations directly underneath it. The diagram is set up to provide two different routes between the thick lines. Now we can use Hess' Law and find two different routes around the diagram which we can equate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
